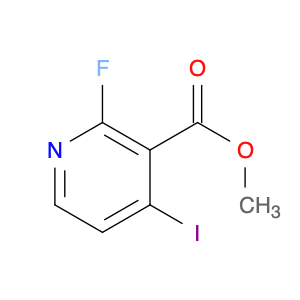
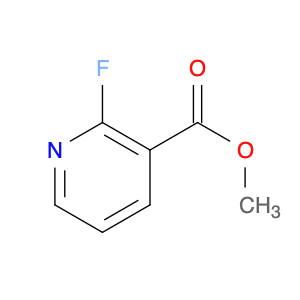
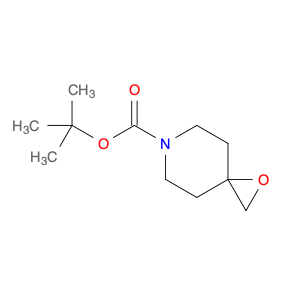
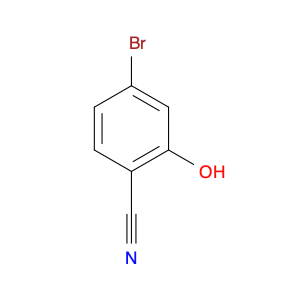
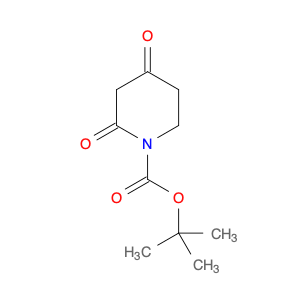
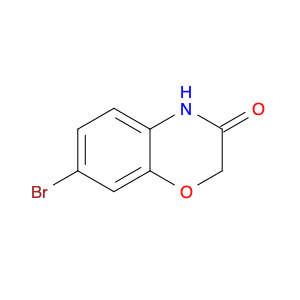
Methyl 2-fluoro-4-iodonicotinate serves as a valuable building block in modern chemical synthesis, especially in the realm of pharmaceutical research and development. This compound plays a crucial role in the creation of novel drug candidates, offering unique properties that can significantly impact the design and efficacy of potential therapeutic agents.In chemical synthesis, Methyl 2-fluoro-4-iodonicotinate is often utilized as a key intermediate in the construction of complex molecular structures. Its strategic placement of both fluorine and iodine functional groups on the nicotinate scaffold imparts distinct reactivity and selectivity profiles, enabling chemists to introduce diverse functionalities into target molecules with precision.The 2-fluoro-4-iodonicotine motif presents an intriguing combination of electron-donating and electron-withdrawing characteristics, which can influence the overall physicochemical properties and biological activities of the final compounds. By harnessing these unique features, researchers can fine-tune the pharmacological profile of potential drug candidates, enhancing their affinity, potency, or metabolic stability.Moreover, the presence of a methyl ester group in Methyl 2-fluoro-4-iodonicotinate offers versatility in synthetic transformations, enabling facile manipulation of the molecule's structure through various chemical reactions. This flexibility allows for the introduction of diverse substituents or functional groups, facilitating the exploration of structure-activity relationships and the optimization of drug-like properties.Overall, the application of Methyl 2-fluoro-4-iodonicotinate in chemical synthesis extends beyond its role as a simple building block, representing a powerful tool for the strategic design and construction of innovative molecular entities with potential therapeutic relevance.
 sales@aaronchem.com
sales@aaronchem.com