Login |
Create New Account
 sales@aaronchem.com
sales@aaronchem.com
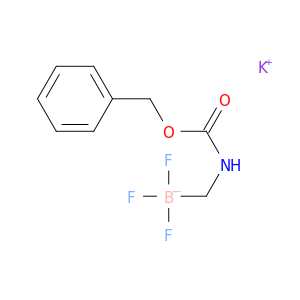
Potassium (benzyloxycarbonylamino)methyltrifluoroborate
Catalog#: AR00AD24 | CAS#: 1695529-70-4 | MDL#: MFCD19982304 | MF: C9H10BF3KNO2 | MW: 271.0857096
| Packsize | Purity | Price | Availability | Quantity | ||
|---|---|---|---|---|---|---|
| 100mg | $41.00 | Global Stock | Buy Now | Add To Cart | ||
| 250mg | $70.00 | Global Stock | Buy Now | Add To Cart | ||
| 500mg | $116.00 | Global Stock | Buy Now | Add To Cart | ||
| 1g | $174.00 | Global Stock | Buy Now | Add To Cart | ||
| 5g | $468.00 | Global Stock | Buy Now | Add To Cart | ||
| 10g | $779.00 | Global Stock | Buy Now | Add To Cart | ||
| 25g | $1,731.00 | Global Stock | Buy Now | Add To Cart |
- Description
- Application
- Related Products
- Featured Products
| Catalog Number | AR00AD24 |
| Chemical Name | Potassium (benzyloxycarbonylamino)methyltrifluoroborate |
| CAS Number | 1695529-70-4 |
| Molecular Formula | C9H10BF3KNO2 |
| Molecular Weight | 271.0857096 |
| MDL Number | MFCD19982304 |
| SMILES | F[B-](CNC(=O)OCc1ccccc1)(F)F.[K+] |